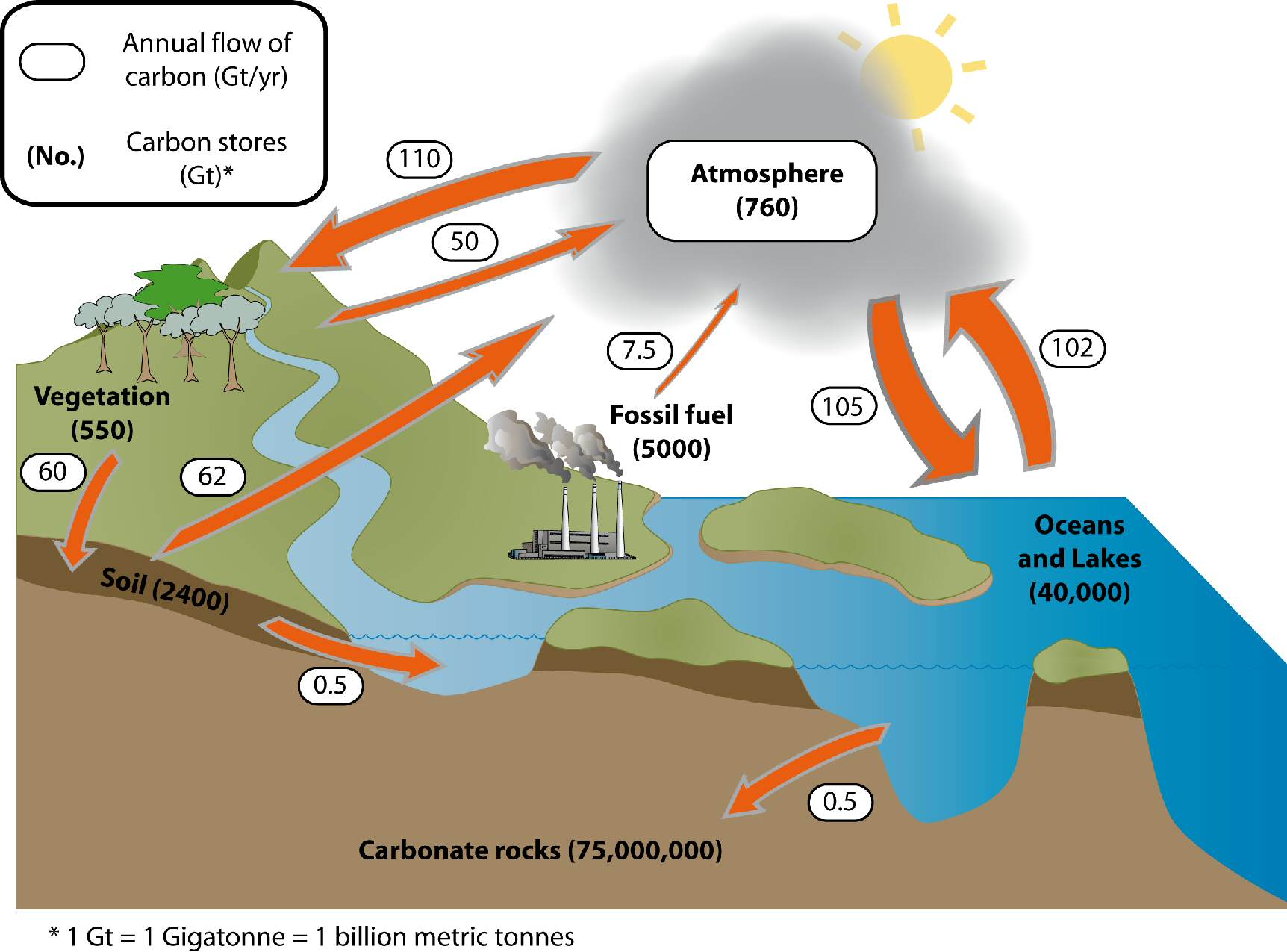
The biosphere is capable of storing ~10% of atmospheric carbon at any given time. Otherwise, anaerobic respiration occurs and releases methane into the surrounding environment, which eventually makes its way into the atmosphere or hydrosphere. When oxygen is present, aerobic respiration occurs, which releases carbon dioxide into the surrounding air or water. Large quantities of carbon pass between the atmosphere and biosphere on short time-scales: the removal of atmospheric carbon occurs during photosynthesis, following the reaction CO 2 + H 2O = CH 2O + O 2, while most carbon leaves the biosphere through respiration, a reversal of the previous reaction in which an amount of energy equivalent to that absorbed during photosynthesis is released as heat. The most important autotrophs for the carbon cycle are trees in forests and phytoplankton in the ocean. This lifestyle requires an external source of energy, for example, the absorption of solar radiation in the process called photosynthesis, or the exploitation of chemical energy sources in a process called chemosynthesis. Autotrophs are organisms that produce their own organic compounds using carbon dioxide from the air or water they live in. It plays an important role in the structure, biochemistry, and nutrition of all living cells. Carbon in the BiosphereĬarbon is an essential part of life on Earth. These minerals dissolve after the death of the organism, but some of the material settles to the sea floor where it can be buried and stored in the form of limestone. Marine organisms build their skeletons and shells out of the minerals calcite and aragonite (CaCO 3) through the incorporation of bicarbonate ions. Carbonic acid dissociates to form bicarbonate ions (HCO 3 -), the form in which most of the carbon in the oceans exists lesser amounts of carbon exist as carbonic acid (H 2CO 3 or dissolved CO 2), and carbonate ions (CO 3 2-) paired with calcium and magnesium and other cations. When carbon (CO 2) enters the ocean, carbonic acid is formed by the reaction: CO 2 + H 2O = H 2CO 3. Carbon is released to the atmosphere at oceanic upwelling sites, whereas regions of downwelling transfer carbon from the atmosphere to the ocean. Inorganic carbon is readily exchanged between the atmosphere and ocean, exerting an important control on the pH of ocean water. release of water vapor, carbon dioxide, and sulfur dioxide from volcanic eruptions and metamorphism.release of dissolved CO 2 back to the atmosphere through warming of surface ocean water and.cement production, when limestone (calcium carbonate) is heated to produce lime (calcium oxide), a component of cement, and CO 2 is released.organic matter combustion (including deforestation and burning fossil fuels), which oxidizes carbon-producing CO 2.decay by fungi and bacteria that break down carbon compounds in dead animals and plants and convert the carbon to CO 2 if oxygen is present, or methane if not.plant and animal respiration, an exothermic reaction that involves the breakdown of organic molecules into CO 2 and water.the reaction between carbonic acid and silicate rock that leads to the production of bicarbonate ions, which are carried to the ocean and eventually deposited in marine carbonate sediments.Ĭarbon can be input to the atmosphere by a variety of processes, including:.the conversion of reduced carbon to organic tissues or carbonates to hard body parts, such as shells, by marine biota in ocean surface waters and.the formation of carbonic acid as circulating ocean surface waters cool near the poles, thereby absorbing more atmospheric CO 2.the photosynthetic conversion of carbon dioxide into carbohydrates by plants, releasing oxygen in the process.The overall atmospheric concentration of these greenhouse gases has increased during the past century and contributed to global warming.Ĭarbon is removed from the atmosphere through: Because CO 2 is chemically inert, it is relatively well mixed within the atmosphere away from forest canopies, whereas CH 4 is chemically active and is removed quickly from the atmosphere through oxidation to CO 2 and water. Most atmospheric carbon is in the form of CO 2, while CH 4 is present only in trace concentrations. The carbon cycle is of interest to understanding climate because it includes two of the most important greenhouse gases: carbon dioxide (CO 2) and methane (CH 4).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
